Mutant 4: Animals and Ocean

Mystique, the shape changer
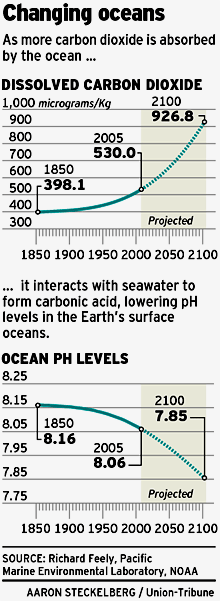
Facts about OceansThe oceans cover 71% of the Earth's surface and contain 97% of the Earth's water. Less than 1% of Earth's water is fresh water, and 2-3% is contained in glaciers and ice caps. The pH of the ocean, like that of human blood, depends on the ratio of bicarbonate to carbon dioxide. Most of the carbon dioxide (CO2) converts to carbonic acid (H2CO3), which dissociates to bicarbonate (HCO3-) and hydrogen ion (H+). The hydrogen ions lower the pH (negative log10(H+)). In 1850, the ocean was at pH 8.16. Today, it is pH 8.11. By the year 2100, it is predicted to be pH 7.85. Coral reefs and other marine phototrophs have shells of calcium carbonate (CaCO3). The shells dissolve as pH decreases. At the deepest point in the ocean the pressure is more than 8 tons per square inch, or the equivalent of one person trying to support 50 jumbo jets. At 39 degrees Fahrenheit, the temperature of almost all of the deep ocean is only a few degrees above freezing. If the ocean's total salt content were dried, it would cover the continents to a depth of 5 feet. |
 |
| Honor
Pledge. I affirm that I have completed this assignment on my own,
aside from the professor and the MSSC. Any outside source of information
must be credited. 1. Mystique's challenge: Suppose you have my shape-changing ability, and you can take the shape of one of our near-human relatives: chimpanzee, gorilla, or bonobo. You could test a question about primate behavior. Which form would you take, and what question would you test? 2. Tribbles evolve with DNA change at 2 % change per ten years. The following new tribble species have evolved (Table). (a) Based on this table, draw a phylogenetic tree showing the relative divergence over time (time shown on the x axis). (b) Explain why the Fribble is included, and what information it provides. Table: Percent Relatedness of Tribble Species
3. Explain what happens to oxygen levels (partial pressure) when: (b) Inhaled air exchanges oxygen with the capillaries, forming 80% oxyhemoglobin. (c) The person with 80% oxyhemoglobin starts to smoke a cigarette
(see handout). 4. In human blood, if the bicarbonate/CO2 ratio is 35,
what is the blood pH? What medical problem does this cause? (a) Calculate the concentration of H+ in the water. (b) Is this pH good for the "raft corals"? Explain why or why not. 6. Suppose a factory releases only 0.0001 moles/liter of hydrogen ions into the Earth's ocean. Will this tiny amount affect the ecosystem? Explain.
|