IV. Serotonin Binding
At the current time, it has only been
possible to crystalize the serotonin
receptor when
which has a
aromatic ring structure as serotonin.
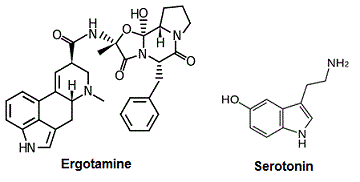
Unsurprisingly, the 5-HT1B receptor has many of
the same interactions with serotonin as it does
with ERG. The Asp129 once
again forms a
with amino side group of serotonin, while Thr134
retains a
with the indole N-H hydrogen of serotonin. This hydrogen
bond is highly conserved in the binding of
serotonin to all receptor sites.
In addition to ERG and serotonin, the serotonin
binding site binds several other aromatic
molecules. LSD binds to 5-HT receptors and is a
potent antagonist at 5-HT7A receptor. Triptans,
such as donitriptan and eletriptan, also fit into
the promiscuous 5-HT receptor binding site.
Norfenfluramine, which is the active metabolic
part of the “Fen-Phen” antiobesity cocktail, also
binds specifically in the orthosteric binding
pocket of the 5HT1B receptor where it forms a
with Phe217 and Tyr218.
V. Future Studies
The interactions between ERG and the
have thus been well classified. With the continuing
improvements made in the field of crystallography
every year, crystalizing serotonin
in the 5-HT1B receptor will provide more
information about the native interaction of
serotonin within the body. Further research will
look into the interactions between all the 5-HT
receptors and the serotonin neuromodulator.
Understanding the interactions of serotonin, not
only the in 5-HT1B receptor, but also in
the
5-HT receptors, will be useful in future medical
studies.
VI. References
Clark MS, Neumaier JF.
(2001). The 5-HT1B receptor: behavioral
implications. Psychopharmacol Bulletin
35(4): 170-85.
Hoyer D, Clarke DE,
Fozard JR, Hartig PR, Martin GR, Mylecharane
EJ, Saxena PR, Humphrey PP. (1994).
International Union of Pharmacology
classification of receptors for
5-hydroxytryptamine (Serotonin). Pharmacological
reviews 46(2): 157-203.
Villalón CM, De Vries P,
Rabelo G, Centurión D, Sánchez, López A, Saxena
P. (1999). Canine external carotid
vasoconstriction to methysergide, ergotamine
and dihydroergotamine: role of 5-HT1B/1D
receptors and 2-adrenoceptors. British
journal of pharmacology 126(3): 585-594.
Wang C, Jiang Y, Ma J, Wu
H, Wacker D, Katritch V, Han GW, Liu W, Huang
XP, Vardy E, McCorvy JD, Gao X, Zhou XE,
Melcher K, Zhang C, Bai F, Yang H, Yang L,
Jiang H, Roth BL, Cherezov V, Stevens RC, Xu
HE. (2013). Structural basis for molecular
recognition at serotonin receptors. Science
340(6132): 610-614.
Return to Top