Homo sapiens Transketolase
Julia Green '20 and Jo Stanley '20
Contents:
I. Introduction
Human transketolase (TKT) is an enzyme involved in the non-oxidative phase of the pentose phosphate pathway. This pathway is responsible for taking ribose 5-phosphate (R5P) and xylulose 5-phosphate and transforming them into fructose 6-phosphate and glyceraldehyde 3-phosphate (G3P), which are substrates used in glycolysis. TKT expression within tissue is consequently linked to central metabolic function; the higher the TKT expression, the higher the metabolism within that tissue. Because of this, TKT has been suggested to be a critical determinant for many biological processes, from the natural, female fertility, to those involved in disease, for example hepatocarcinoma and fibromyalgia. This is possibly due to the high metabolic rate involved in these processes.
The human genome also codes for two transketolase-like proteins, TKTL1 and TKTL2. While these proteins likely have a function similar to that of TKT, their exact biological function is unknown. TKTL1 has been tentatively linked to carcinogenesis, as there is a strong correlation between the presence of TKTL1 within a tissue and its susceptibility to cancer cell invasion. The function of TKTL2 remains mysterious.
II. General Structure
Approximately fifty amino acids are conserved across all species, and many of these conserved amino acids are involved in cofactor and substrate binding this will be discussed more in Section IV. Activating Regions.
Biologically, human TKT usually exists as a homodimer; however, when crystallized, it formed a monomer, so the structure shown here is one subunit of a homodimer. Human TKT has three different domains : the N-terminal PP domain that contacts the pyrophosphate region of ThDP; Pyr domain that binds the aminopyrimidine part of ThDP; C-terminal domain, who�s biological function is currently unknown in the scientific community. These three distinct domains are connected through flexible linker regions
between the PP & Pyr domains, as well as between the Pyr & C-terminal domains.
The same residues that contact the alpha-phosphate and the beta-phosphates of the ThDP, also contact the Ca2+ ion . However, the residues that bind to the beta-phosphate of the ThDP also interacts with a water molecule as well as the Ca2+ ion .
Human (and other mammalian) TKT lacks an active histidine that is conserved in all non-mammalian TKTs. This His481 is replaced with an active Gln428 instead, this is highlighted when the button is clicked. Before the active Gln428 was discovered in mammalian TKTs, the theory for the function of the active His481 was that it was used as an acid/base catalyst. However, this hypothesis was disproved when the active His481 was not found in mammalian TKTs. The new theory for the function of these active centers in TKTs is that the residue helps with substrate binding.
III. Mechanism
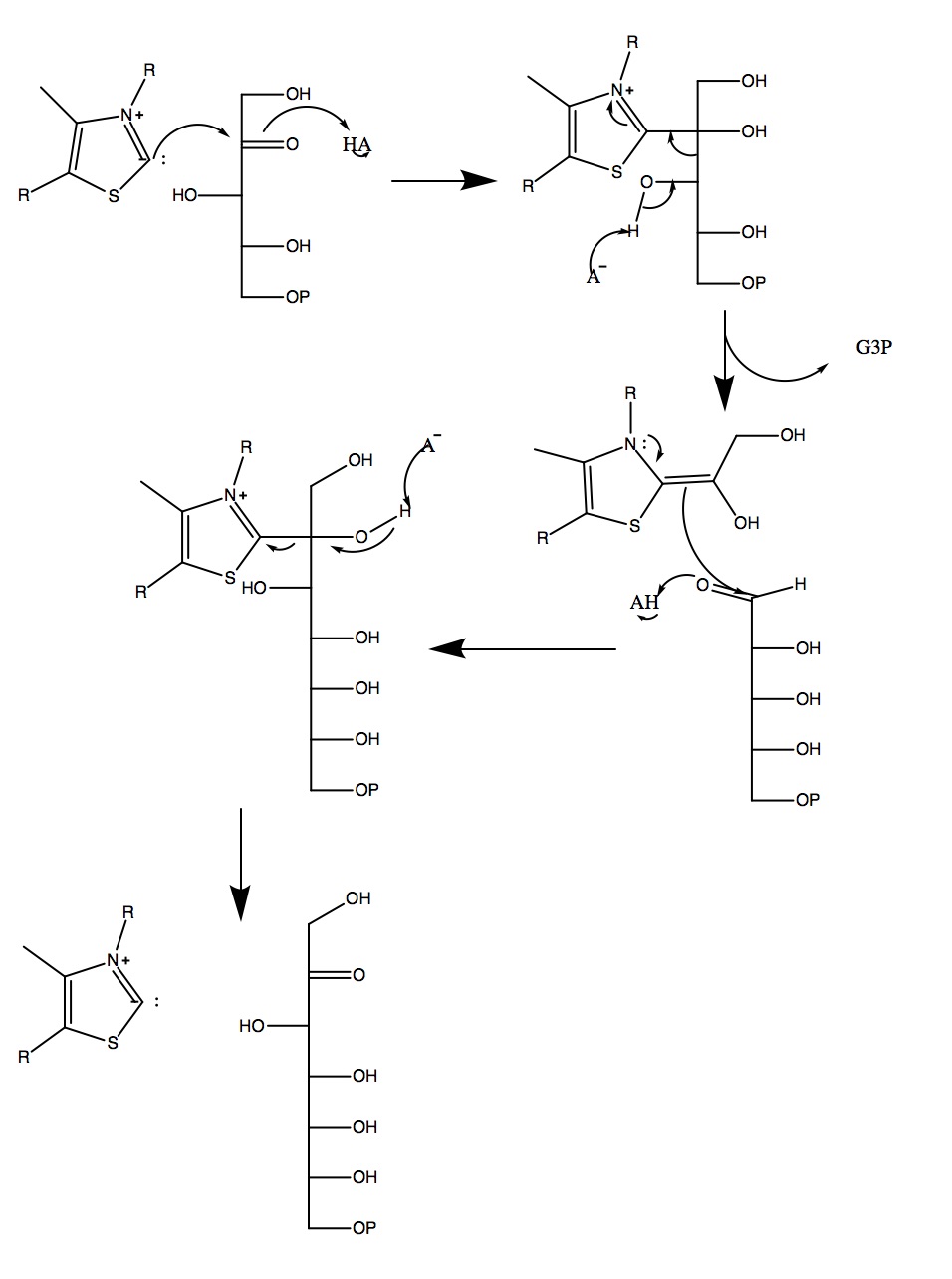
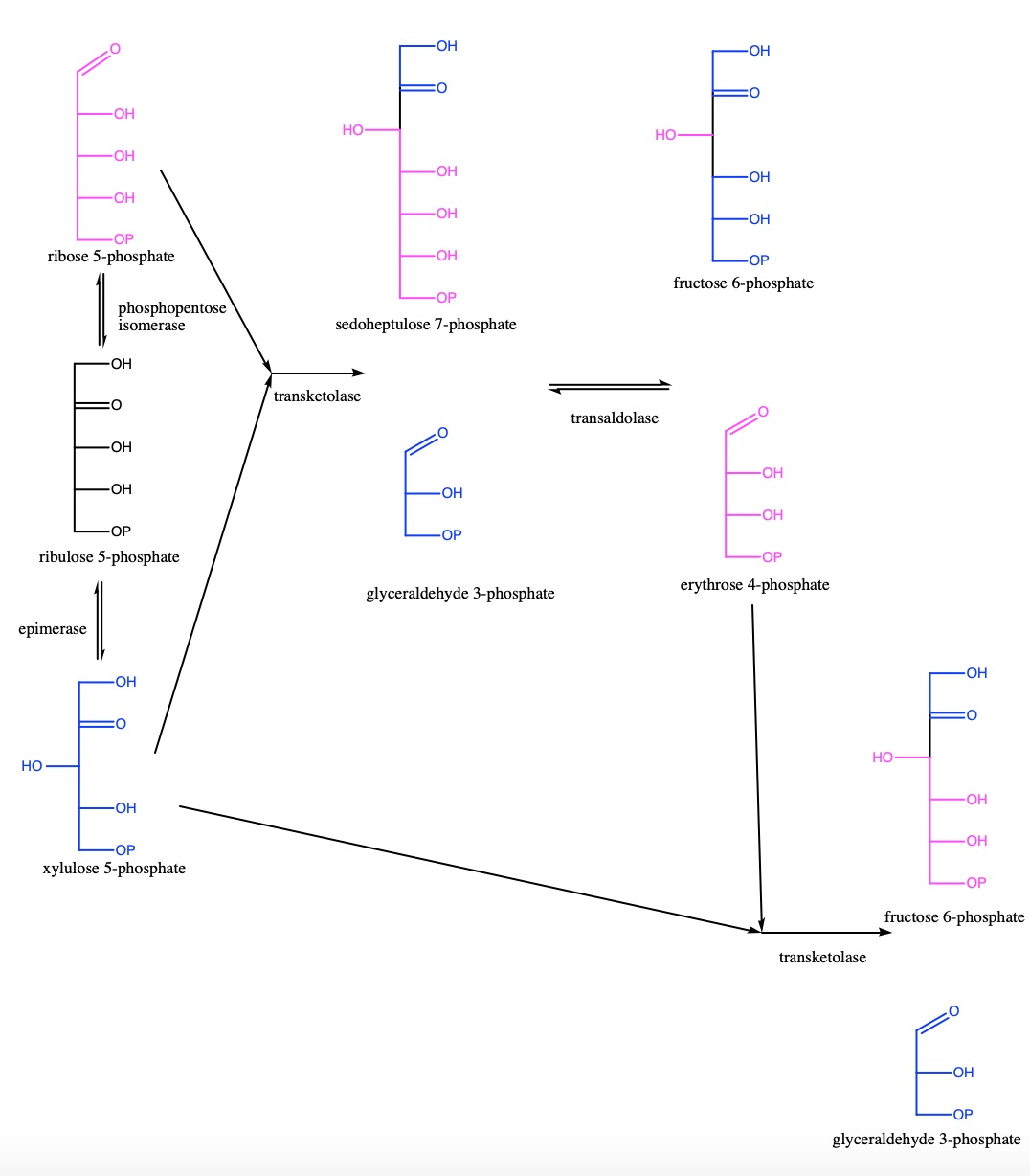
Human TKT plays a
vital part in the non-oxidative phase of the pentose phosphate
pathway, and it acts on specific phosphate sugars to transform them
into sugars that can be incorporated in glycolysis. It binds a
cofactor called thiamine pyrophosphate (TPP or ThDP), which holds on
to the intermediates during the reaction. TKT is used for two
different reactions within the pentose phosphate pathway; in the
first, it uses ribose 5-phosphate and xylulose 5-phosphate to
produce glyceraldehyde 3-phosphate and sedoheptulose 7-phosphate.
Although glyceraldehyde 3-phosphate is a substrate for a glycolytic
enzyme, it usually does not leave the pentose phosphate pathway to
enter into glycolysis at this time. Instead, transaldolase will take
the products of the first TKT reaction and use them to produce
fructose 6-phosphate (which will
leave to enter glycolysis) and erythrose 4-phosphate. The second TKT
reaction takes the erythrose 4-phosphate and xylulose 5-phosphate to
produce fructose 6-phosphate and glyceraldehyde 3-phosphate, both of
which will then enter into glycolysis.
IV. Activating Regions
Active site with the non-covalently bound cofactors ThDP and Ca2+ is found at the dimer interface between the N-terminal PP domain and the Pyr domains of both subunits of TKT. ThDP is primarily held in place by hydrogen bonding and hydrophobic interactions between the ThDP, the TKT protein, and the water molecule. The Ca2+ ion also contacts the TKT protein as well as the water molecule, in fact all of the residues that bind with the Ca2+ also bind with water. These can be seen when the button is clicked.
Many of the active sites are conserved in both human and E. coli TKT, and are found in incredibly similar positions. An example of this can be seen when the button is clicked.
V. References
Mitschke, L., Parthier, C., Schroder-Tittmann, K., Coy, J., Ludtke, S., Tittmann, K. (2010) J. Biol. Chem. 285: 31559-31570.
Back to Top