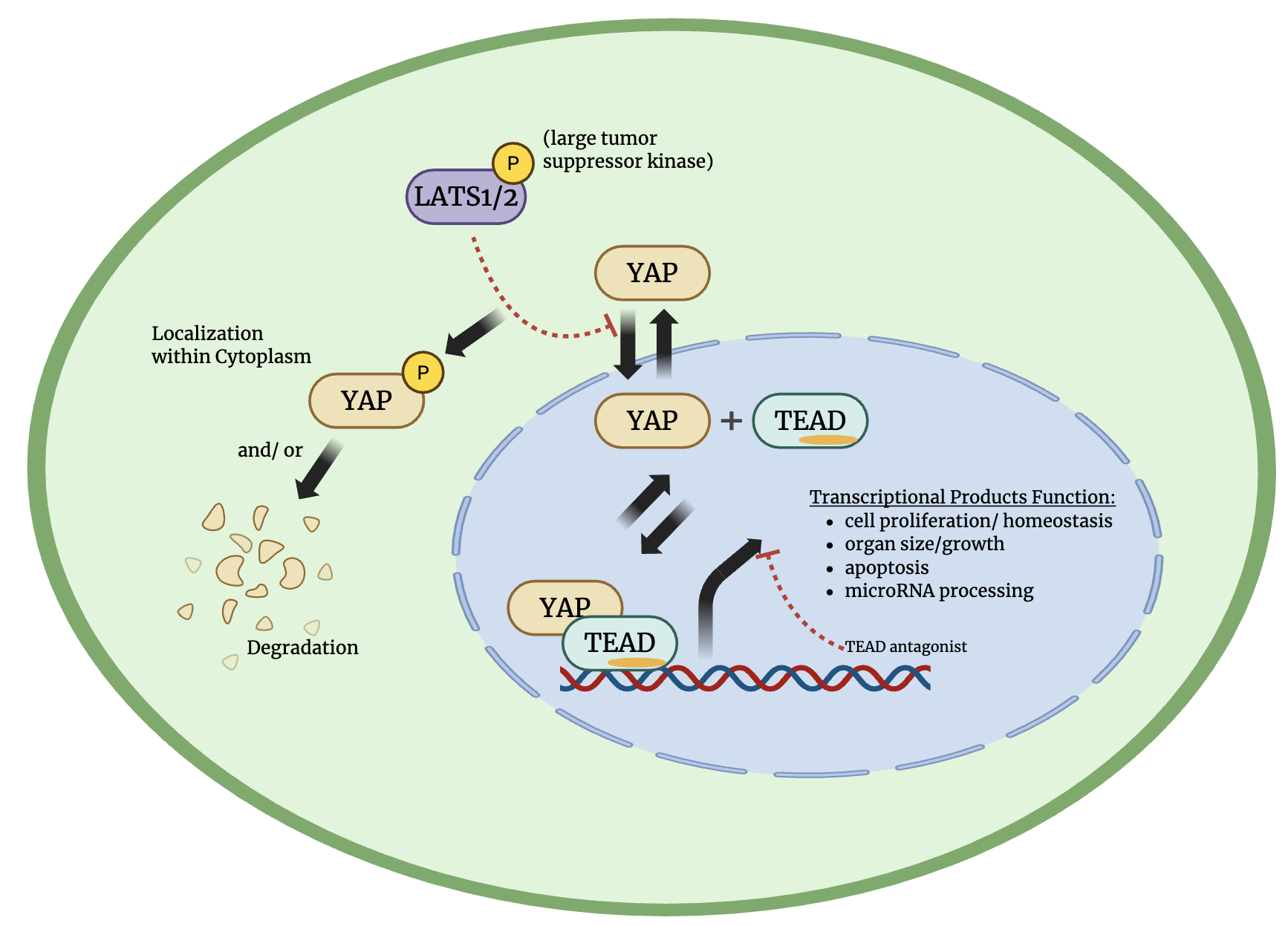
 Figure 1: Simplified overveiw of the Hippo Tumor Suppressor Pathway. The green circle represents a cell with the blue dotted circle representing the nucleus. Figure made by Dominic Simpson in BioRender, inspiration taken from Holden and Cunningham (2018).3
Figure 1: Simplified overveiw of the Hippo Tumor Suppressor Pathway. The green circle represents a cell with the blue dotted circle representing the nucleus. Figure made by Dominic Simpson in BioRender, inspiration taken from Holden and Cunningham (2018).3
The Yes-associated protein (YAP) is a transcriptional coactivator known for inducing cell proliferation, homeostasis, and organ size/growth, while also regulating transcription factors involved in apoptosis and microRNA processing. YAP is key protein that is regulated through the Hippo tumor suppressor pathway, which when active suppresses YAP's 'ability to translocate to the nucleus and perform the functions listed above.6
YAP has been observed to function as a candidate oncogene for several reasons, two key ones being: 1) its overexpression in various cancers and 2) impaired cell proliferation of YAP inhibited cells.1,8
The Transcriptional Enhanced Associate Domains (TEAD 1–4) are a family of transcription factors that recruit YAP binding in the nucleus through the
C-terminal transactivation domain and bind DNA through their
N-terminal TEA domain (Figure 2).5
TEAD is the main transcription factor involved in YAP-induced growth and oncogene functioning, making it crucial for the cell proliferation process.9
Similar to YAP, possesses oncogenic functions, as it is heavily involved in human malignancies and is considered a key driver in the initiation and progression of cancer.10 YAP’s ability to bind to TEAD occurs only when the Hippo pathway is inactivated; the resulting YAP-TEAD complex is what stimulates gene expression. Because of the oncogenic roles of both YAP and TEAD, the ability to manipulate the YAP-TEAD complex has become an important area of research for anticancer therapies and gaining a deeper understanding of Hippo pathway regulation.5
![Binding Domains]() Figure 2: Binding domains of YAP and TEAD. Numbers indicate amino acids. Image produced by Li et al. (2010).4
Figure 2: Binding domains of YAP and TEAD. Numbers indicate amino acids. Image produced by Li et al. (2010).4
2. General Structure
The YAP-TEAD complex forms a
although the two heterodimers here form a dimerised version prodiced in a single unit cell during x-ray crystallography (resolution: 2.80 Å).
YAP consists of an ⍺-helix and a 𝝱-strand
, and a very key coil region( 𝛀-loop)
which is considered to be intrinsically disordered until conformationally changed and stabilized when bound to TEAD.5,7
TEAD is composed of four ⍺-helices that wrap around a 𝝱-sandwich (12 beta-strands. packed against each other).
YAP encompasses TEAD1’s globular structure using three differentially essential interfaces: YAP 𝝱1 and TEAD1 β7,
YAP's ⍺1 and TEAD1’s ⍺3/4,
YAP’s 𝛀-loop and TEAD1’s 𝝱4, 𝝱11, 𝝱12, ⍺1, and ⍺4.
In mammals, the TEAD binding domain of YAP is highly conserved, which suggests an evolutionarily conserved partnership between YAP and TEAD.5 YAP-TEAD4 complex formation is a one step mechanism though YAP ⍺1 folds and binds to TEAD4 independent from, and before 𝛀-loop formation; suggestive of a diffusion-collision protein folding mechanism.2
This binding mechanism is potentially the same here for YAP-TEAD1 since TEADs 1-4 in humans contain very conserved residues and bind similarly to YAP; though TEADs 1-4 have slightly differentiated functions related to tissue type.2,5,7
3. Interface Binding
is mediated by several hydrogen bonds from the peptide backbones of YAP 𝝱1 and TEAD 𝝱7. Residues 52-58 on YAP form these bonds with TEAD residues 318-324, producing antiparallel 𝝱-sheets. Of the three interfaces, YAP’s 𝝱1’s proves to be the most variable in its sequence across species as there are limited side chain interactions.
In vitro GST-TEAD pull down assays show the deletion of YAP 𝝱1 causing little change to the binding of TEAD; while the CTGF reporter assay of the same deletion caused a modest decrease in binding affinity. Both assays are suggestive of a minor role of interface one in binding the YAP-TEAD complex.5
is mediated by hydrophobic interactions between the LXXLF motif of YAP ⍺1 with the hydrophobic groove made by TEAD ⍺3/4. More specifically, the hydrophobic interactions of this region include: YAP L65 with TEAD Y346, F350 and K353, YAP L68 with F314, F350 and F370, YAP F69 with TEAD F350, K353, L354, L357, and V366, and finally the three YAP residues with each other. Similar to interface one, In vitro GST-TEAD pull down assays of mutated residues in the LXXLF motif: L68A and F69A did not affect YAP-TEAD binding.
The functional CTGF reporter assay showed that L68A mutation in YAP causes a similar decrease in binding affinity as the 𝝱1 deletion in YAP.5
The 𝛀-loop of YAP mediates
by sliding side chain residues into a groove made by TEADs 𝝱4, 𝝱11, 𝝱12, ⍺1, and ⍺4. Multiple
interactions are observed between YAPs M86, L91, and F95 with TEADs L247, V242, L272, V391, and Y406 .
strengthen this interaction and are formed through: YAPs guanidinium group of R89 with TEADs carboxylate group of D249, YAP S94 with TEADs Y406 and E240. Internal Hydrophobic interactions of YAP: F96, M86, R87, L91, and F95 stabilizes the coil region.
YAPs P98 and P99 are stabilized by with TEADs W276 and H404.
Both the GST-TEAD pull down assay and CTGF reporter assay for YAP mutations: M86A, R89A, L91A, S94A, F95A, and F96A were proven to cause the most dramatic decreases in TEAD1 binding affinity; emphasizing the structural importance of interface three on complex formation. Mutagenesis experiments in the YAP-TEAD4 complex also identified the 𝛀-loop of YAP as a “hot spot” for complex interactions.2,5
4. References
1. Avruch, J., Zhou, D., & Bardeesy, N. (2012). Yap oncogene overexpression
supercharges colon cancer proliferation. Cell Cycle, 11(6),
1090–1096. https://doi.org/10.4161/cc.11.6.19453
2. Bokhovchuk, F., Mesrouze, Y., Meyerhofer, M., Zimmermann, C., Fontana, P., Erdmann, D., Jemth, P., & Chène, P. (2020). An early association between the α-helix of the TEAD binding domain of yap and Tead drives the formation of the yap:tead complex. Biochemistry, 59(19), 1804–1812. https://doi.org/10.1021/acs.biochem.0c00217
3. Holden, J., & Cunningham, C. (2018). Targeting the hippo pathway and cancer through the Tead family of transcription factors. Cancers, 10(3), 81. https://doi.org/10.3390/cancers10030081
4. Kang, W., Tong, J. H. M., Chan, A. W. H., Lee, T.-L., Lung, R. W. M., Leung, P. P. S., So, K. K. Y., Wu, K., Fan, D., Yu, J., Sung, J. J. Y., & To, K.-F. (2023). Data from Yes-Associated Protein 1 Exhibits Oncogenic Property in Gastric Cancer and Its Nuclear Accumulation Associates with Poor Prognosis. https://doi.org/10.1158/1078-0432.c.6519390.v1
5. Li, Z., Zhao, B., Wang, P., Chen, F., Dong, Z., Yang, H., Guan, K.-L., & Xu, Y. (2010). Structural insights into the yap and tead complex. Genes & Development, 24(3), 235–240. https://doi.org/10.1101/gad.1865810
6. Sebio, A., & Lenz, H.-J. (2015). Molecular pathways: Hippo signaling, a critical tumor suppressor. Clinical Cancer Research, 21(22), 5002–5007. https://doi.org/10.1158/1078-0432.ccr-15-0411
7. Tian, W., Yu, J., Tomchick, D. R., Pan, D., & Luo, X. (2010). Structural and functional analysis of the YAP-binding domain of human TEAD2. Proceedings of the National Academy of Sciences, 107(16), 7293–7298. https://doi.org/10.1073/pnas.1000293107
8. Tsujiura, M., Mazack, V., Sudol, M., Kaspar, H. G., Nash, J., Carey, D. J., & Gogoi, R. (2014). Yes-associated protein (YAP) modulates oncogenic features and radiation sensitivity in endometrial cancer. PLoS ONE, 9(6). https://doi.org/10.1371/journal.pone.0100974
9. Zhao, B., Ye, X., Yu, J., Li, L., Li, W., Li, S., Yu, J., Lin, J. D., Wang, C.-Y., Chinnaiyan, A. M., Lai, Z.-C., & Guan, K.-L. (2008). Tead mediates yap-dependent gene induction and Growth Control. Genes & Development, 22(14), 1962–1971. https://doi.org/10.1101/gad.1664408
10. Zhou, Y., Huang, T., Cheng, A., Yu, J., Kang, W., & To, K. (2016). The Tead family and its oncogenic role in promoting tumorigenesis. International Journal of Molecular Sciences, 17(1), 138. https://doi.org/10.3390/ijms17010138
Back to Top