CB1 Receptor in Complex with Synthetic Ligand AM6538
Lucas Macy '28 and Spencer Stluka '28
Contents:
I. Introduction
The CB1 receptor belongs to the family of proteins called G protein coupled receptors (GPCR) and is the most highly expressed GPCR in the human brain. It is additionally expressed throughout the body, with high levels also found in the central nervous system. The CB1 receptor is part of what is called the endocannabinoid system, a network of receptors that mediates downstream signaling, which in turn regulates many different processes, including memory, pain, and appetite, making it a valuable target for many types of drug therapies.
The CB1 receptor is best known for its role in cannabis consumption, as it can bind THC as a ligand, the psychoactive component of cannabis. However, the CB1 receptor can bind to many different ligands, both endogenous and exogenous. For ease of crystallization, a synthetic ligand AM6538 was used in order to stabilize the crystal structure.
II. General Structure
As mentioned earlier, the structure of CB1 was determined using AM6538 as a ligand, a strategically modified version of rimbonant. AM6538 was used because it stabilizes the bound complex, enhancing the ability to form useful crystals. The wild-type CB1 receptor was also modified, also to assist crystal formation. It was truncated on both the N- and C- termini by 98 and 58 residues respectively. Additionally, four computationally predicted mutations for expression and thermostability were introduced into the CB1 receptor (Thr210Ala, Glu273Lys, Thr283Val, Arg340Glu). The CB1 receptor contains seven transmembrane ɑ-helices (helix I,II,III,IV,V,VI,VII,), which are connected by three extracellular loops (ECL1-3,),
three intracellular loops (ICL1-3) and an amphipathic helix (VIII). ECL2 consists of 21 residues that are folded into a structure that projects four residues into the binding pocket. These four residues are important for mediating interactions with certain classes of ligands. Specifically the two cysteines (Cys 257 and Cys 264) are critical for the function of CB1. The two cysteine also constrain the conformation of ECL2 by the presence of an interloop disulfide bond.
III. Ligand Structure
AM6538 (Figure 1) was synthesized through strategic modifications of rimonabant to enhance its ability to stabilize the ligand–receptor complex and promote CB1 crystal formation. Unlike rimonabant, its 5-phenyl ring substituent was altered to introduce structural motifs that increase affinity for the CB1 receptor. AM251, a rimonabant analog and well-established CB1-selective antagonist, served as the precursor for AM6538’s synthesis. The synthetic strategy involved functionalizing the para-iodo substituent on the 5-phenyl ring of AM251 with an acetylenic four-carbon chain bearing a substituent at the terminal carbon. At the fourth carbon of the alkyne unit, a nitrate group (ONO₂) was introduced to act as a polar leaving group that could be displaced by a suitable nucleophile within or near the binding domain; alternatively, the nitrate could remain intact, forming a near-irreversible, non-covalent attachment via interactions with hydrogen-bonding residues and residues capable of π–π interactions.
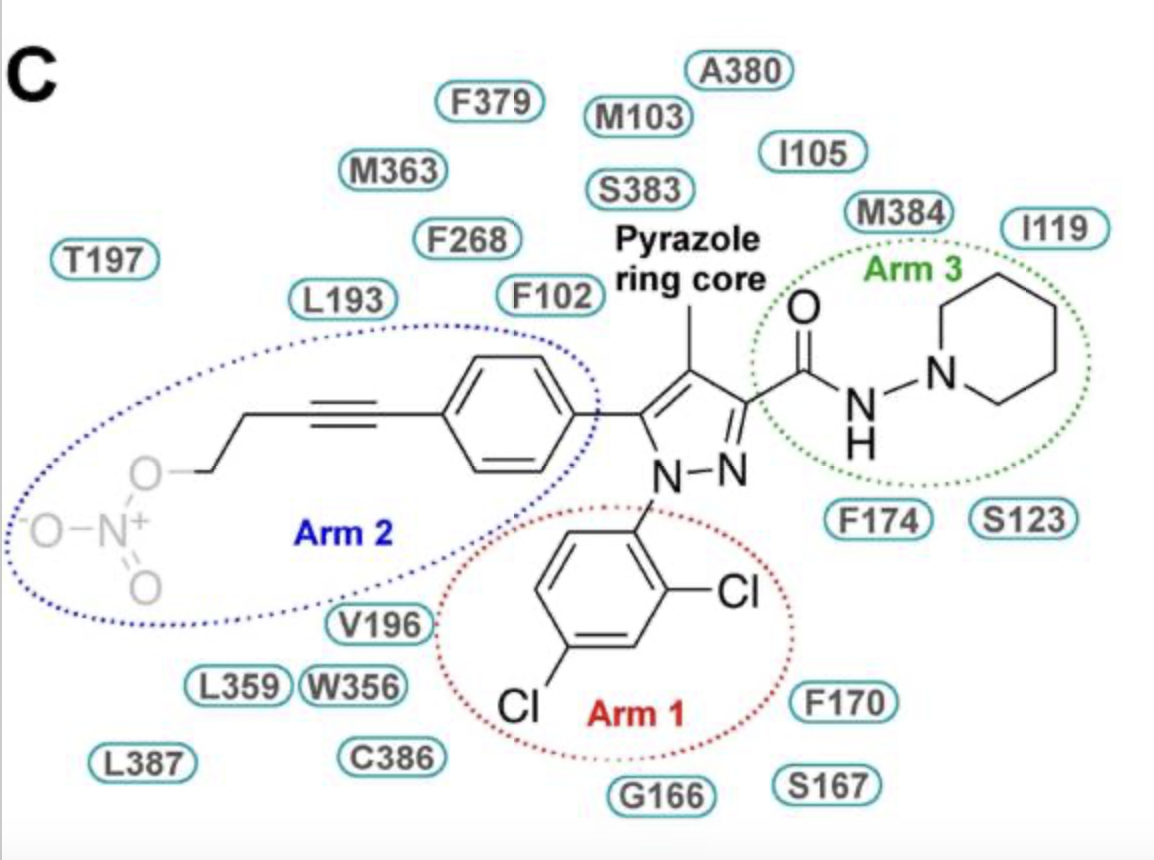
Figure 1: Structure of AM6538
IV. Ligand Binding Pocket
AM6538 lies unusually low within the CB1 binding pocket compared to other orthosteric ligands of class A GPCRs and primarily forms hydrophobic interactions. The non-truncated N terminus forms a V-shaped loop (residues 99–112) that inserts into the binding cavity and contributes additional stabilizing interactions. Its
is positioned bearing three functional groups between helices II and VII, where it forms hydrophobic interactions with Phe170, Phe379, and Ser383 and is capped by the N-terminal loop. The ligand engages primarily in hydrophobic contacts with ECL2, the N terminus, and all helices except helix IV.
occupies a narrow side pocket formed by helices II, III, VI, and VIII, where its 2,4-dichlorophenyl ring forms edge-face π–π interactions with Phe170 and the Gly166–Ser167 backbone, along with hydrophobic contacts with Val196, Trp356, Cys386, Leu387, and Met103; the excellent fit of the dichlorophenyl group explains why 2,4-dichloro or 2-chloro substitutions optimize binding.
extends into a long narrow channel shaped by helices III, V, VI, and ECL2, where its phenyl group forms π–π interactions with Phe102, Phe268, and Trp356, along with hydrophobic interactions involving Leu193, Val196, and Leu359; the positioning of this long hydrophobic chain resembles that of ML056 in the S1P receptor, suggesting a conserved lipid-binding channel.
projects toward a gap made by helices I, II, and VII and capped by the N-terminal loop, forming predominantly non-specific hydrophobic interactions with Met103, Ile105, Ile119, Ser123, Phe170, Phe174, Ala380, Ser383, and Met384.
V. Medical Applications
The CB1 receptor is part of the endocannabinoid system (ECS), which mediates many important bodily functions such as learning, memory, emotions, sleep, temperature control, pain, inflammatory and immune responses, and appetite. The CB1 receptor is one of the most common receptors in the brain, and it acts to control the release and activity of most other neurotransmitters in the brain, which gives rise to the wide range of its effects on the body. Endocannabinoids are types of endogenous ligands, such as anandamide, which the body produces naturally, but there are also other exogenous ligands, such as THC and rimonabant.
Likely the best known exogenous ligand of the CB1 receptor is THC, the primary psychoactive component of cannabis, has several established and emerging medical uses, largely tied to its ability to impact neuronal signaling through CB1 receptors in the central nervous system. Clinically, THC is used to deal with chronic pain, nausea and vomiting due to chemotherapy, and stimulating appetite in people with AIDS anorexia.
THC acts as a partial agonist of CB1 receptors, which are G protein coupled receptors abundantly expressed in brain regions involved in pain perception, appetite regulation, memory, and motor control. THC's effects on nausea, appetite, and pain are attributed to its binding of CB1 receptors in the CNS, which modulate sensory, somatic, and cognitive perception. When THC binds to CB1, it stabilizes receptor conformations that inhibit adenylate cyclase, decrease cAMP production, modulate ion channels, and reduce neurotransmitter release including glutamate and GABA thereby dampening neuronal excitability. This mechanism underlies both its therapeutic effects, such as nausea relief, and analgesia, and its psychoactive effects, including altered perception and cognition.
Rimonabant is an antagonist of the CB1 receptor and was developed as an anti-obesity drug, due to the hypothesis that since THC stimulates appetite as an agonist of CB1, rimonabant should suppress appetite as an antagonist. Although it was successful in this, due to the many functions of the ECS, it also caused anxiety, depression, and suicidal thoughts which led it to be pulled from the market. As mentioned earlier, a modified version of rimonabant was used in determining the crystal structure of CB1 in complex with a ligand, with the notable change on arm 2, replacing the 4-chlorophenyl ring on rimonabant with a 4-aliphatic chain substituted phenyl ring on AM6538.
Due to its wide range of effects on the body, the ECS and the CB1 receptor are being targeted in drug discovery to treat pain, obesity, mental illness, neurodegenerative disorders, and more. Understanding the structure of CB1 and how it binds to different ligands aids in this process, and helps scientists develop new drugs that can hopefully mitigate unwanted side effects.
VI. References
Hua T, Vemuri K, Pu M, Qu L, Han GW, Wu Y, Zhao S, Shui W, Li S, Korde A, Laprairie RB, Stahl EL, Ho JH, Zvonok N, Zhou H, Kufareva I, Wu B, Zhao Q, Hanson MA, Bohn LM, Makriyannis A, Stevens RC, Liu ZJ. Crystal Structure of the Human Cannabinoid Receptor CB1. Cell. 2016 Oct 20;167(3):750-762.e14. doi: 10.1016/j.cell.2016.10.004. PMID: 27768894; PMCID: PMC5322940. Grinspoon, Peter. “The Endocannabinoid System: Essential and Mysterious.” Harvard Health, 11 Aug. 2021, www.health.harvard.edu/blog/the-endocannabinoid-system-essential-and-mysterious-202108112569.
Ng T, Gupta V, Keshock MC. Tetrahydrocannabinol (THC) [Updated 2023 Nov 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK563174/
Lan R, Liu Q, Fan P, Lin S, Fernando SR, McCallion D, Pertwee R, Makriyannis A. Structure-activity relationships of pyrazole derivatives as cannabinoid receptor antagonists. J Med Chem. 1999;42:769–776. doi: 10.1021/jm980363y.
Picone RP, Khanolkar AD, Xu W, Ayotte LA, Thakur GA, Hurst DP, Abood ME, Reggio PH, Fournier DJ, Makriyannis A.(−)-7’-Isothiocyanato-11-hydroxy-1’,1’-dimethylheptylhexahydrocannabinol (AM841), a high-affinity electrophilic ligand, interacts covalently with a cysteine in helix six and activates the CB1 cannabinoid receptor. Mol Pharmacol. 2005;68:1623–1635. doi: 10.1124/mol.105.014407.
Back to Top