Structure of the HoxB1-Pbx1 Heterodimer Bound to DNA
Natasha Jose '27 and Dylan Ryznar '28
Contents:
I. Introduction
The HoxB1 protein is a member of the Hox homeodomain proteins which are important transcription factors, regulating development and directing the body plan of many organisms. All Hox genes have a highly conserved, 60 amino acid DNA binding domain, so the individual proteins enhance their sequence specificity through interactions with a cofactor, in this case Pbx1. Pbx1 was originally discovered as a proto-oncogene that becomes disrupted by the process of chromosomal translocation, which occurs in approximately 25% of pre-beta cell leukemias (often in children). Hox proteins were initially discovered in Drosophila fruit flies, but they have very similar orthologs in vertebrates; HoxB1, found in vertebrates, is orthologous to the Drosophila labial protein, which is an important regulator of head and midgut development. Additionally, the cofactor Pbx1 in vertebrates is orthologous to the extradenticle (exd) cofactor in Drosophila. The formation of HoxB1-Pbx1 heterodimer is extremely important, as it is essential for head and facial development in vertebrates, specifically relating to the formation of the VII cranial nerve, the innervation of various facial muscles, and hindbrain segmentation.
II. General Structure
The entire structure of the HoxB1 and Pbx1 proteins was not elucidated, but instead only the minimal domains of the proteins required for high affinity cooperative binding to DNA. The heterodimerization and general interactions of HoxB1 with Pbx1 occurs by way of a
which is found at the N-terminus of the HoxB1 DNA-binding domain, the conserved sequence being YFPWMKR, and the HoxB1-specific sequence being TFDWMK (residues −24 to −19). This hexapeptide is attached to the Hox homeodomain through a disordered flexible linker. The consensus sequence of this linker is variable amongst different Hox proteins in different species; the exact physiological significance of this variability is not yet known. The HoxB1 hexapeptide binds to Pbx1 through a , formed by the space between a three-residue insertion characteristic of Pbx1, the C-terminal 310 helix and helix 3 of the Pbx1 homeodomain. It is important to note that the C-terminal residues of the Pbx1 homeodomain contribute to the stabilization of the hexapeptide binding pocket, but they do not directly contact the HoxB1 homeodomain, as the HoxB1 and Pbx1 DNA-binding sites are on opposing sides of the DNA helix.
The tryptophan in the HoxB1 hexapeptide forms van der Waals interactions with several Pbx1 residues in the homeodomain including phenylalanine-20 in helix 1; leucine-23a, proline-24, tyrosine-25; and arginine-53, tyrosine-56, and lysine-57 on helix 3.
III. Pbx1 Structure
Pbx1 is a member of TALE (three-amino-acid loop extension), which is a family of homeodomain proteins that is unique due to a particular characteristic: a
at the C terminus of the homeodomain of helix 1. The DNA binding of Pbx1 is unique among homeodomain proteins due to the fact that, in addition to the traditional 3 helices of the homeodomain, it also contains a fourth helix that packs against the homeodomain. Residues 60-72 of Pbx1 form a 310 helix, followed by a two-turn α helix () that instead of directly interacting with HoxB1 or the DNA, packs against helix 1 and helix 3 of the Pbx1 homeodomain, forming a stabilized, four-helix DNA- binding domain.
The four-helix DNA-binding domain is characterized by many intramolecular chemical interactions, including but not limited to van der Waals interactions,
, and hydrophobic interactions. For example, in the bend following helix 3, Ile-60 is involved in van der Waals interactions with Tyr-56 of helix 3, and Tyr-70 is involved in van der Waals interactions with both Asn-17 and Glu-18. Additionally, hydrogen bond formation mediated by a water molecule is exemplified by the interactions between Tyr-56 and the reactive carbonyl group of Ile-60. Further hydrogen bonds form between Glu-66 and Tyr-21,and between Tyr-70 and Glu-14. Hydrophobic interactions, exemplified by the contacts between Ala-67 (in helix 4) and Tyr-21 (helix 1 residue), also play an important role with respect to the chemical composition of the four-helix DNA-binding domain. In addition, Glu-66 in helix 4 forms a
with Lys-52 in helix 3 and a hydrogen bond with Tyr-21 in helix 1.
IV. DNA Binding
HoxB1 and Pbx bind to opposite sides of the same 11 bp sequence, reading 5’-ATGATTGATCG-3’. Both proteins form sequence specific contacts with the DNA bases and sugar-phosphate backbone.
HoxB1 Binding
Asn-51 of HoxB1 forms two with A8 of the DNA binding region, a contact that is conserved throughout most homeodomain proteins. Gln-50 forms an additional
hydrogen bond with C11 on the opposite strand. Ile-47, Met-54, and Gln-50 also form van der Waals contacts with specific bases. Finally, bound solvent water molecules mediate further hydrogen bonds between HoxB1 residues and specific DNA bases in the sequence.
Pbx1 Binding
The binding domain of Pbx1 is similar to that of HoxB1, because they are both homeodomains. That being said, Pbx1 has more sequence specificity than HoxB1. Pbx1 has the same Asn-51 hydrogen bonds, these with A4. The Arg-55 residue also forms two hydrogen bonds with G3. Asn-47 and Ile-54 form van der Waals contacts with T5 and A6, and Pbx1 also has a water mediated network of hydrogen bonds allowing for further contacts between the binding domain and the DNA.
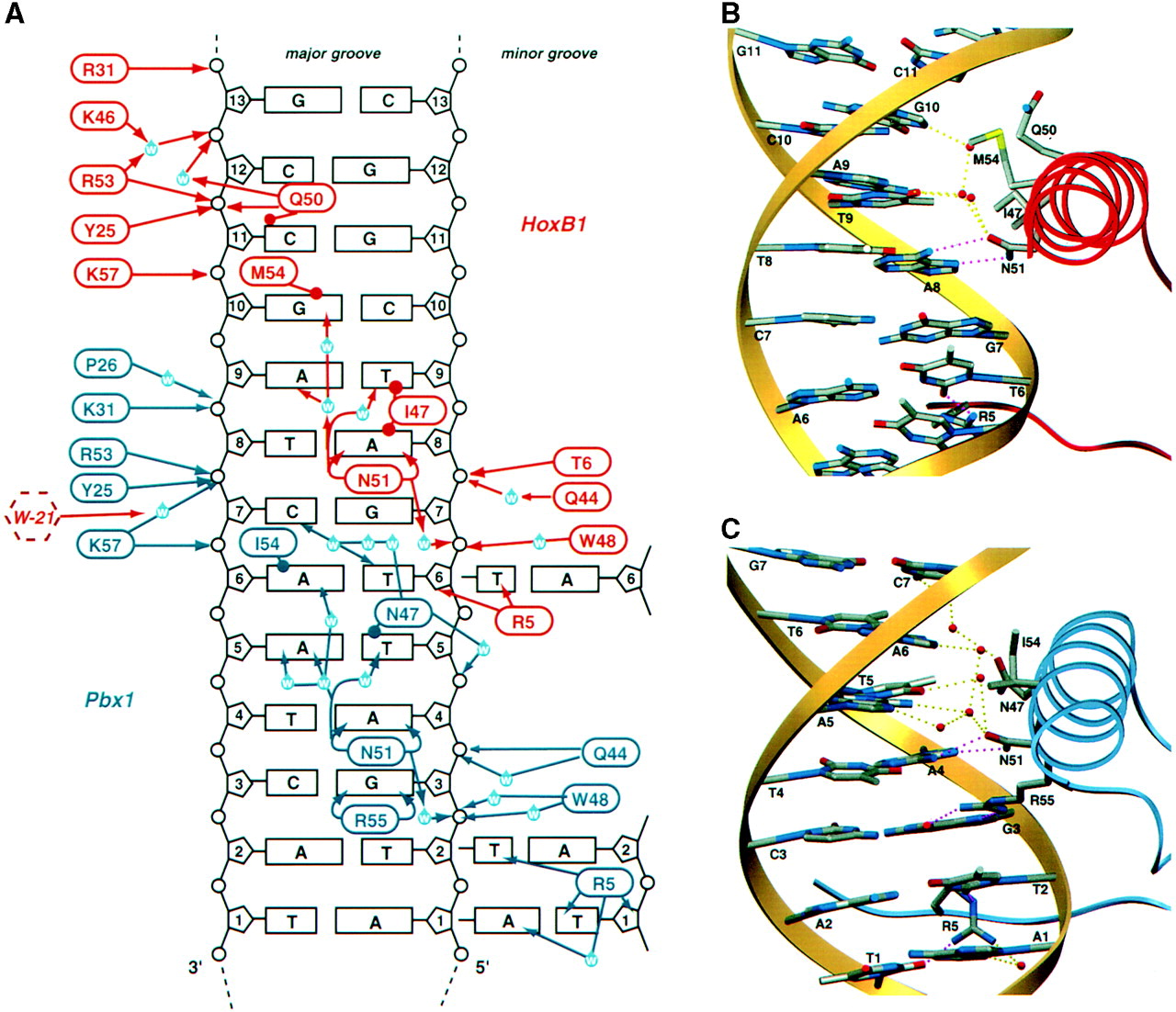
Figure 1. Diagram of interactions between the HoxB1-Pbx1 heterodimer and the DNA sequence the complex selectively binds to (Slupsky et al).
V. Mutations
A study conducted by Webb et. al (2012) focused on the effects of HoxB1 mutations on the facial development of humans. The researchers identified German-American children who had a homozygous missense mutation in the highly conserved Arg-5 of the HoxB1 homeodomain, resulting in Arg-5 being substituted by a cysteine. Arg-5 contributes to enhanced DNA binding particularly through interactions with thymine in the minor groove of DNA, facilitated by hydrogen bonding and electrostatic attraction, as its characteristic guanidino group is positively charged at physiological pH. Cysteine lacks a positively charged functional group and has a comparatively reduced hydrogen-bonding capacity when compared with Arg-5, which results in weaker DNA contacts. As this mutation results in destabilization of the HoxB1-DNA complex, it alters HoxB1 transcriptional activity, leading to the formation of disorders such as Congenital Facial Paralysis (CFP). This demonstrates the importance of minor-groove chemical interactions with respect to the stabilization of HoxB1-DNA binding required for cranial system development.
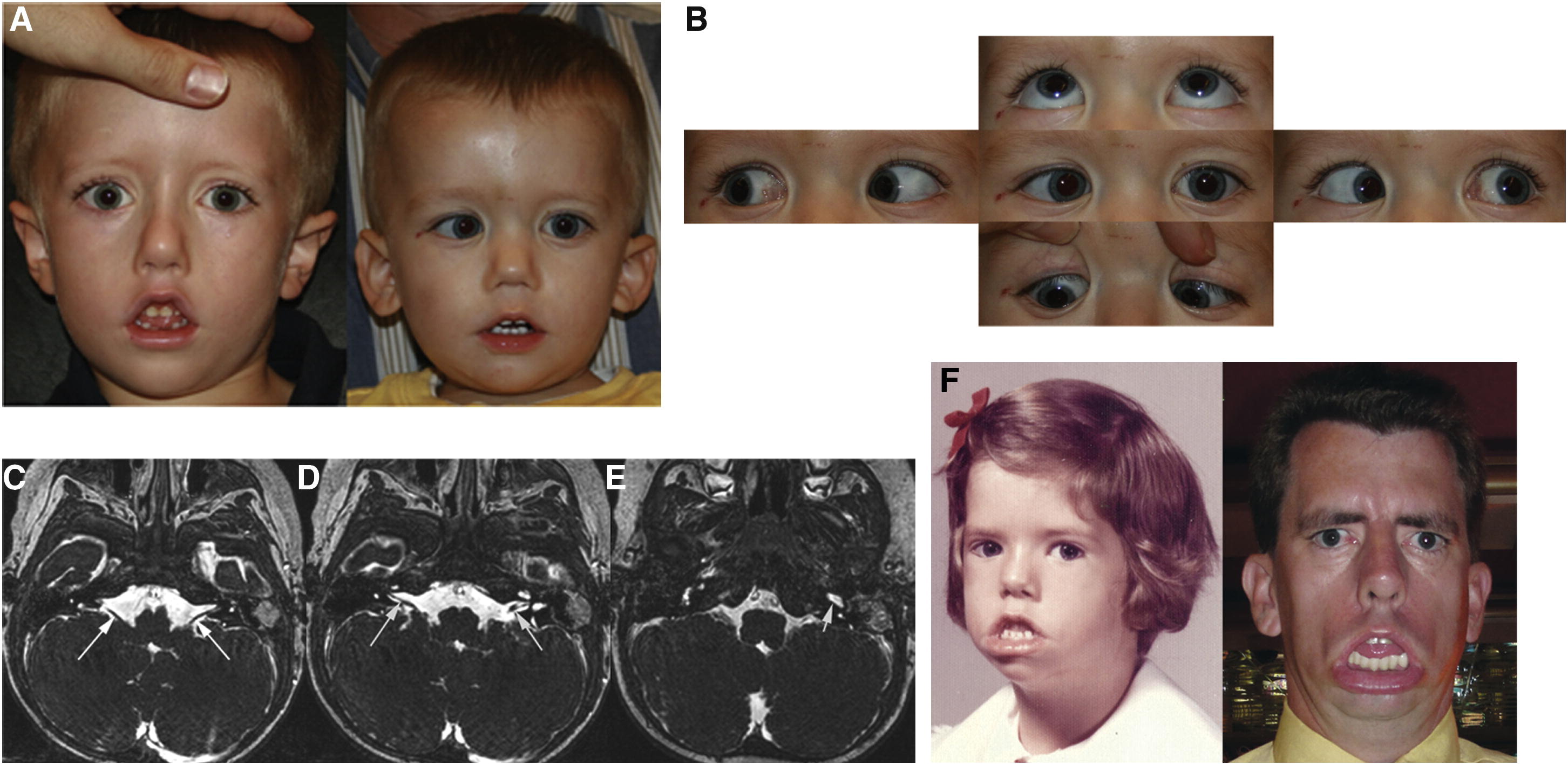
Figure 2. Individuals with the HoxB1 missense mutation.
VI. Conclusion
The HoxB1-Pbx1 heterodimer complex forms as a result of chemical interactions between the HoxB1 and Pbx1 proteins. The HoxB1 protein interacts with the Pbx1 protein through a hexapeptide chain located on the N-terminal side of the HoxB1 DNA-binding domain. The hexapeptide chain interacts with Pbx1 through a unique binding pocket formed by the space between the three-residue insertion of Pbx1, as well as the C-terminal 310 helix and helix 3 of the Pbx1 homeodomain. The Pbx1 protein is a unique part of the TALE family, as it contains a fourth helix in addition to the traditional 3 helices of the homeodomain that packs against the homeodomain and contributes to the formation of the DNA-Pbx1 binding pocket. Specifically, residues 60-72 of Pbx1 are responsible for the formation of a 310 helix, and a two-turn α helix (helix 4) that pack against helix 1 and helix 3 of the Pbx1 homeodomain, resulting in the formation the four-helix DNA- binding domain. The formation of the HoxB1-Pbx1 heterodimer is essential for body plan (specifically cranial system) development, as the binding of the Pbx1 homeodomain protein with the HoxB1 protein contributes to enhanced DNA sequence specificity.
VII. References
Lomberk, Gwen A et al. “Conservation of the TGFbeta/Labial homeobox signaling loop in endoderm-derived cells between Drosophila and mammals.” Pancreatology : official journal of the International Association of Pancreatology (IAP) ... [et al.] vol. 10,1 (2010): 74-84. doi:10.1159/000276895
Slupsky, C M et al. “The HoxB1 hexapeptide is a prefolded domain: implications for the Pbx1/Hox interaction.” Protein science : a publication of the Protein Society vol. 10,6 (2001): 1244-53. doi:10.1110/ps.50901
Webb, Bryn D., et al. "HOXB1 founder mutation in humans recapitulates the phenotype of Hoxb1−/− mice." The American Journal of Human Genetics 91.1 (2012): 171-179.
Back to Top