

Fritz Went first found Auxin in 1926 by isolating the compound that causes coleoptile tip bending towards light. It is now understood that Auxin contributes to growth of leaves, flowers, fruits and stems of grasses and conifers. Most of this evidence comes from experiments involving the manipulation of exogenous Auxin levels. It is probably that developmental changes are caused by changes in the frequency or availability of auxin receptor protiens. (Salisbury and Ross, 1992) The exact mechanism involved with the binding of auxin is not know however current research is quickly building an understanding of what is happening.
Some evidence for receptor control over auxin activity comes from a mutant form of tomato called diageotropica (dgt). These dgt mutants contain normal levels of auxin but in some ways behave as if they are auxin defficient. The stems of these plants are diagravatropic in that they grow approximately horizontal. The stems fail to respond to added auxin either by faster ethylene production or faster elongation of excised hypocotyl sections (Kelly and Bradford, 1986). These dgt mutatants appear to lack an auxin receptor in the endoplamic reticulum (Hicks et al, 1989). In wildtype maize ABP has been localized within the lumen of the ER and on the outer surface of the plasma membrane by silver enhansed immunogould labeling (Diekman, Venis, and Robinson, 1995). One experiment reported anti-idiotopic localization of ABP in the nucleus of a wide range of plants and claimed that this might give evidence for ABP involvment in regulation of gene activity (Prasad and Jones, 1991).
Previous experiments on receptors of Auxin have centred on isolation of mutants and the development of molecular tools to analyze these mutants. More current research involves the application of tools like viral and eukaryotic expression cloning. The ABP in maize and strawberry has been recenly solubilized, characterized, cloned and sequenced. In maize the ABP (indirectly by sequensing a DNA that codes for it) is a dimer made of two polypeptides aboud 20kDa each. This protien has been expressed in insect cells via the baculovirus expression system (MacDonald et. al, 1994). This expressed ABP Is localized in the ER of insect cells in accordance with its signal peptide sequence and appears to be active in binding auxin homologues (MacDonald, et al., 1994). Other recent research on ABP include the cloning into mamalian cells to find the putative ER-ABP docking protien. (www.mpiz...). 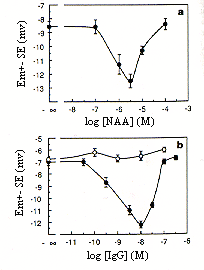
Figure 3. Dose response curves of the Em of tobacco mesophyll protoplasts to NAA (a) and to preimmune (o) and antipeptide D16 (filled circles) antibodies (b). Protoplasts were isolated from wild-type tobacco plants. Mean Em values were obtained for each experimental condition from 15 (a) or 20(b) individual measurements, and the corresponding SE values were calculated.
Antibodies specifically constructed to recognize a polypeptid portion of ABP have located what appears to be an essential portion of the auxin binding site. These antibodies cause auxi-like activity such as hyperpolarization of protoplast transmembrane potential (see figure 3) (Venis et al, 1992). It appears that the antipeptide D16 antibodies cause a similar depolarization of the membrane that the auxin analogue NAA causes. This is evidence for the antipeptide binding in the ligand binding site of the ausin receptor because the binding causes a hormone-like responce.
It appears that multiple receptors for the auxin signal could exist in each cell allowing a complexity of signal perception mechanisms to exist maybe even involving other signal hormones.

 Congradulations you finished! Now return to the introduction and pick another topic!
Congradulations you finished! Now return to the introduction and pick another topic!
